Clinical Neuroscience Laboratory
of Sex Differences in the Brain
Pioneering discoveries of sex differences in disorders of the brain and comorbidity with general medicine
&
Development of novel sex-selective therapies targeted to brain & heart
“Through discovery, translation, education,
we are revolutionizing
the way
in which
the world understands the relationships
between
the brain and heart
disorders
using sex and gender lenses”
Founder and Executive Director: Jill M. Goldstein, PhD, MPH
Dr Jill M. Goldstein
-
Dr. Goldstein is a clinical neuroscientist and population scientist, Professor of Psychiatry and Medicine at Harvard Medical School, Founder and Executive Director, Innovation Center on Sex Differences in Medicine (ICON-✘) at Massachusetts General Hospital and the Helen T. Moerschner Endowed MGH Research Institute Chair in Women’s Health.
She is an expert on sex differences in the brain and their implications for understanding sex differences in psychiatric disorders and co-occurrence with general medical disorders. For >30 years, Dr Goldstein has been a trailblazer in the field of sex differences and women’s health studies bringing methodologic rigor and novel ideas about shared pathophysiology in understanding sex differences in disorders of the brain and heart.
Her team consists of an interdisciplinary group of investigators, integrating structural and functional MRI studies, psychophysiology, steroid hormones, markers of immune function, genetics/genomics, and collaborative efforts with preclinical investigators modeling hormones, genes, immune function, and the brain.
She has been funded by NIH for >30 years to support the work, served on scientific advisory boards for women’s health, brain health, and Alzheimer’s disease, and participated in strategic planning for the National Institutes of Health (NIMH & ORWH) and National Academy of Medicine.
-
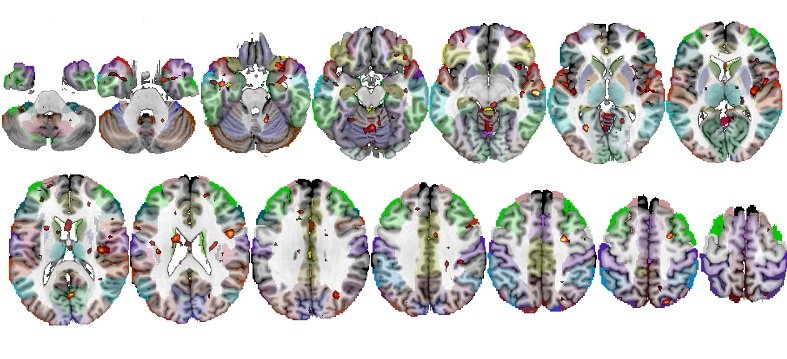
Beginning in the 1980’s, Dr. Goldstein led the way in rethinking the importance of sex in the risk for schizophrenia even though the prevailing view was that it was similar in men and women. Studies demonstrated that men have a significantly higher risk than women, the causes for which began during mid-gestation, a key period of the sexual differentiation of the brain. She published the first MRI study of sex differences across the entire healthy brain >20 years ago, laying the groundwork for how the development of these differences was disrupted prenatally by sex in both schizophrenia and major depression.
Her team went on to uncover how prenatal maternal stress was transmitted to offspring brain development in a sex-dependent way, providing critical insights into understanding early antecedents to the risk for sex differences in psychoses and depression and abnormalities in brain circuitries (stress, mood/anxiety, memory) and physiology (hormonal, autonomic function, and immune) that are shared across disorders of the brain and heart. Her team discovered some of the same early antecedents implicated in depression were shared with risk for cardiovascular disease. In 2021, in the largest psychiatric genetics consortium (PGC) dataset to date, her team uncovered sex differences in genes (shared and non-shared) between depression, schizophrenia, and bipolar psychosis.
Together, this work has contributed to understanding shared pathophysiology that cross disorders of the brain as well as with the heart.
Using a sex differences lens, studies have taken a lifespan approach from fetal development to aging, focusing on periods of reproductive changes. Since 2014, work in the lab has expanded to identify developmental risks for sex differences in memory decline and risk for Alzheimer’s disease in early midlife and the impact of reproductive age as distinct from chronologic age.
Dr Goldstein has published >200 articles, chapters and other original and peer-reviewed work in these areas (+ Info)
-
Dr Goldstein spent her career at Harvard training, mentoring, and sponsoring the next generation in women’s health/sex differences in medicine, including leading for almost 20 years an ORWH Harvard-wide K12 junior faculty training program on building interdisciplinary careers in women’s health.
In 2018, she launched the Innovation Center on Sex Differences in Medicine (ICON-✘) at MGH across departments and divisions, whose mission is to enhance discoveries of sex differences in medicine and translate them into the development of sex-dependent therapeutics to address the need for precision medicine.
“When It Comes to Health, Sex Matters”
Stay tuned for the latest news on Sex-Specific research and discoveries
Follow us at @ICON-X and @JillMGoldstein on Twitter
About Us
The Clinical Neuroscience Laboratory of Sex Differences in the Brain integrates scientists from clinical and basic neuroscience perspectives to address questions of why men and women are at different risks for disorders of the brain and their comorbidity with general medical disorders, such as cardiovascular disease (CVD).
The work of the lab takes a lifespan approach to their investigations targeting naturalistic windows of opportunity for the study of sex differences when the brain and body are differentially flooded or depleted of gonadal hormones, e.g., fetal development and puberty, and, for female-specific periods, pregnancy and the menopausal transition compared to age-matched men.
The work in the lab is particularly focused on the roles of steroid hormones, genes, and markers of immune function in understanding sex effects in major depression, psychoses, memory decline and risk for Alzheimer’s disease and comorbidity of these with cardiometabolic disorders.
We believe that an understanding of these hormonal, genetic, and immune pathways will provide knowledge for the development of sex-dependent and sex-specific diagnostic tools and therapies.
Under Dr Goldstein’s leadership
the Clinical Neuroscience Lab has been focusing on
Sex differences in health and disease
Developing Sex-Selective Novel Therapeutics
Understanding comorbidity of disorders of the brain with general medical diseases, particularly the brain and heart

Innovation Center on Sex Differences in Medicine (ICON-✘)
The CNL-SD sits in the broader context of the Innovation Center on SeX Differences in Medicine (ICON-X) at Massachusetts General Hospital, Harvard Medical School, connecting collaborators across Harvard & other Institutions crossing fields, methodologies and departments.
Founder and Executive Director, Jill M. Goldstein, Ph.D. Massachusetts General Hospital, HMS
Discover
Studies of shared pathophysiology for understanding sex differences in disorders of the brain & heart, e.g., depression, CVD, and Alzheimer’s disease
Collaborate
Build stronger ties between investigators across Harvard system & other academic centers and Innovative firms in the US and globally